April 2026 – Updates on the SCN2A Drug Development Pipeline

Dear SCN2A Community,
Everything starts with a committed and engaged community.
The trust you place in us to share updates, provide context, and help navigate an evolving research landscape is something we value deeply.
Updates about Elsunersen (PRAX-222) ASO Therapy
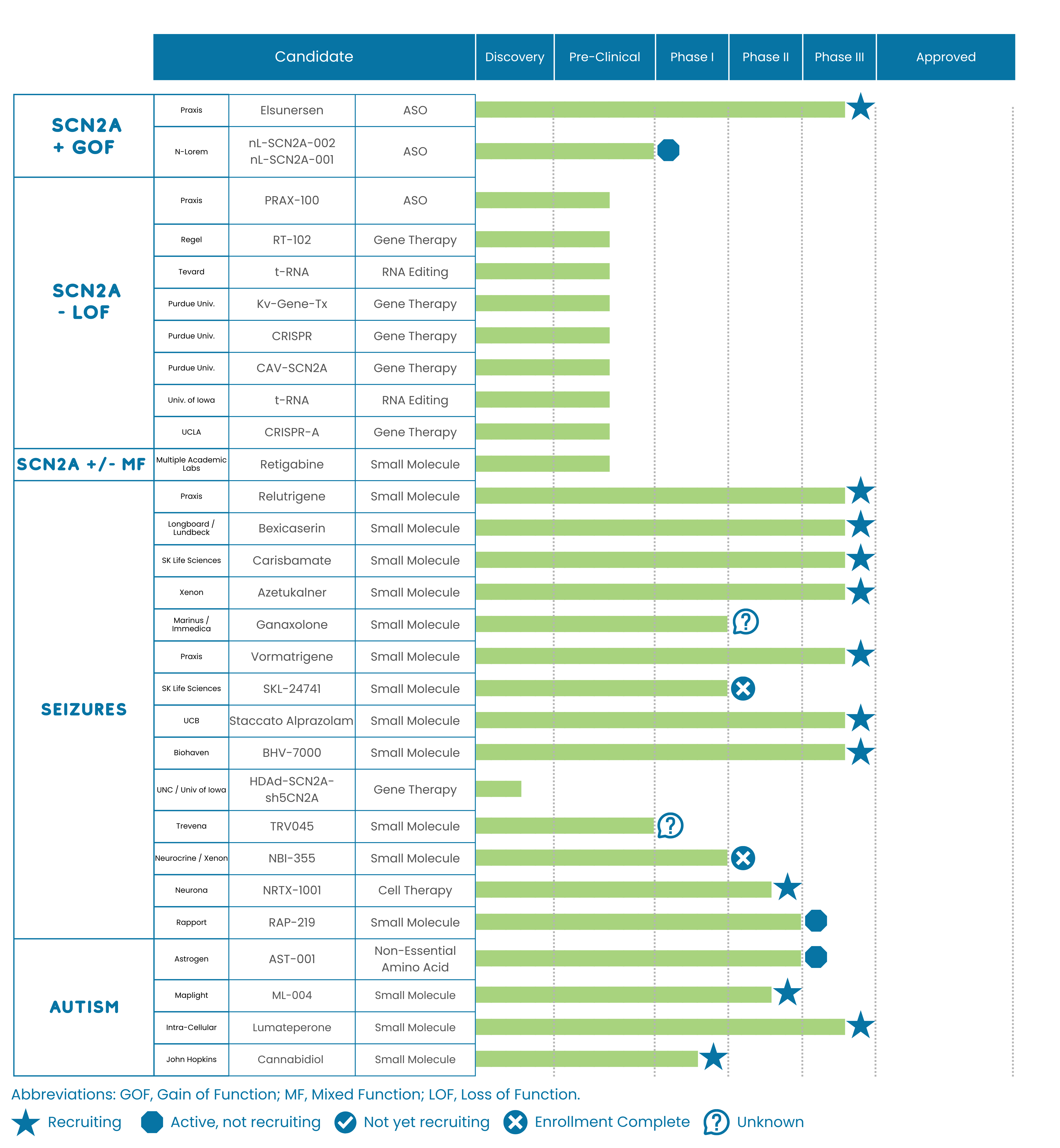
Our team attended a meeting on April 6, 2026, where Praxis updated global SCN2A advocacy leaders about Elsunersen (PRAX-222). The Praxis team shared topline results from the EMBRAVE Part A Study (Phase 1/2), which tested the safety and effectiveness of their Antisense Oligonucleotide (ASO) for Gain of Function variants in SCN2A. In total, there were 9 participants (7 receiving the study ASO and 2 receiving a placebo). The results demonstrated a 77% placebo-adjusted reduction in motor seizures (p=0.0152). This means that the study showed participants who received Elsunersen had approximately 77% fewer motor seizures compared to those who received a placebo. While the results from this early testing show some promising results, it is important to remember that they are based on a small sample of 9 participants and may not reflect responses in all individuals with Gain of Function SCN2A variants. We are waiting for larger Phase 3 study results to understand what role Elsunersen may play in future treatments for individuals with Gain of Function SCN2A variants.
The next step for Elsunersen in the drug development pipeline is a Phase 3 clinical trial called the EMBRAVE3 study. This study is now recruiting in the United States (California, Illinois, Ohio, Colorado, Tennessee, and Pennsylvania) and will also recruit in Europe (Italy, Germany, and the United Kingdom) in the coming weeks. In this study, all participants will receive the study ASO, and no participants will receive a placebo.
As always, we recommend speaking with your medical team to see if participating in a clinical trial like EMBRAVE3 is a good option for your family.
Updates about Relutrigine (PRAX-562)
Additionally, Praxis also released an update on the submission of their New Drug Application (NDA) for Relutrigine (PRAX-562), a small molecule treatment for SCN2A-DEE and SCN8A-DEE to the Food and Drug Administration (FDA). The FDA has accepted Priority Review of the NDA, with a target action date under the Prescription Drug User Fee Act (PDUFA). The FDA is planning to announce an update on its review of the NDA by September 27th, 2026. Importantly, this update does not guarantee approval of the treatment, but serves as a “checkpoint” date to monitor for status updates on the NDA review.
Progress in rare disease happens step by step, and each new result adds to a broader and evolving understanding. It is important to know and understand your loved one’s SCN2A variant as this can directly inform decisions for care management. Given that there is a wide spectrum of symptoms that can present in someone with a change in the SCN2A gene, it is very likely that no one treatment regimen is the same for each individual. The FamilieSCN2A Foundation encourages you to communicate with your medical team regarding any questions about clinical management, treatment regimens, and variant interpretation and classification. For more information and additional educational resources, please visit The Genetics of SCN2A page on our website.
Next Steps
The FamilieSCN2A Foundation remains committed to advancing patient-centered drug development and supporting the broader SCN2A research landscape. As the FDA reviews applications for new treatments for SCN2A, they will rely heavily on information provided by advocacy engagement and research publications. The Foundation continues to fund and support critical research, including registries, natural history studies, endpoint development, and biomarker studies. Participation in natural history studies, including the Dragonfly Study, as well as support for the research that moves science forward, all play a role in what comes next.
One of the strongest roles the FamilieSCN2A Foundation can play is to ensure patient-centered drug development efforts. This includes working with the global SCN2A community to share the critical patient voice with the FDA so that the reviewers understand what is truly meaningful to families. Upcoming advocacy opportunities include hosting an Externally-led Patient-Focused Drug Development (PFDD) meeting with the FDA on October 23rd, 2026. Families from around the world will be invited to share their experiences, which will directly impact the information the FDA uses to evaluate experimental treatments.
We continue to monitor ongoing research and treatment development efforts and share updates with care, context, and a focus on what matters most to this community. For the most current view of SCN2A research and treatment development, we encourage you to visit our Research Roadmap and Treatment Pipeline.
What remains constant is this community
We saw the strength of this community at the start of this year. SCN2A Awareness Day reached further than ever before, driven by families, advocates, clinicians, and partners who chose to show up and share.
It was collective, and it made a real impact.
That kind of momentum doesn’t happen by accident; it’s built over time through connection and trust, and it helps drive the broader work happening across the SCN2A landscape.
Thank you for your continued trust in our foundation. If you have any questions about these updates, we encourage you to reach out to our team.
With gratitude,
FamilieSCN2A Foundation Team
Leah Myers – Executive Director
Jeff Cottrell – Chief Scientific Officer
Amanda Gale – Director of Programs & Operations
Angie Weaver – Director of Philanthropy & Development
Melody Kisor – Director of Advocacy & Education
Morgan Weberg – Research Coordinator
Post authored by: Morgan Weberg – Research Coordinator and Amanda Gale – Director of Programs & Operations